
When considering a substance, following the steps below will help you determine what type(s) of intermolecular forces exist in the substance.Ĭlick on each number to see steps to follow. With these overlapping ranges, how can the intermolecular forces of two different substances be compared? The summary below will answer this question. 
This means a statement such as "Dipole-dipole forces are stronger than London forces" can not be made. 016 - London Dispersion ForcesIn this video Paul Andersen describes the positive force intermolecular forces found between all atoms and molecules. The D4 model is presented for the accurate computation of London dispersion interactions in density functional theory approximations (DFT-D4) and generally. More importantly, the ranges for the various intermolecular forces overlap. * For comparison, the approximate magnitude of covalent bonds is 100-400 kJ/mol.Īs you can see in the table, the strength of each type of intermolecular force covers a range of values. The table below compares the strength of various intermoelcular forces. I learned that what results in London fores between molecules is the 'cloud of electrons' that results in an instantaneous dipole and induced dipole, which eventually results into. Finally, we propose a natural extension to the method to include quadrupolar and octupolar dispersion interactions.While intermolecular forces are strong enough to keep molecules together in the solid and liquid state, they are not nearly as strong as covalent bonds. The predominant intermolecular force in (CH3)2NH is:A) London-dispersion forcesB) ion-dipole attractionsC) hydrogen bondingD) dipole-dipole attractions2) London dispersio. 'London (dispersion) forces are responsible for the fact that non-polar substances can be condensed to form liquids and sometimes solids at low temperatures'. We also show that ab initio correlated methods include dispersion effects naturally and discuss the balanced treatment of dispersion using these techniques. They represent an induced dipole interacting with an induced. This permits computation of instantaneous dispersion in systems with constrained mutual rotation and the formation of dispersion aggregates. London Dispersion forces are pretty clearly explained in Londons paper on intramolecular forces. It exists in all physical states of matter and are universal. Van derwaal’s forces are very weak attractive forces existing between molecules. Il principe William e Kate Middleton stanno progettando di trasferirsi da Londra al Berkshire. The discussion below extends the description of dispersion to all separations and orientations. Answer (1 of 4): London dispersion forces are a type of van derwaal’s forces. I duchi di Cambridge hanno deciso: il loro futuro sarà lontano da Londra.  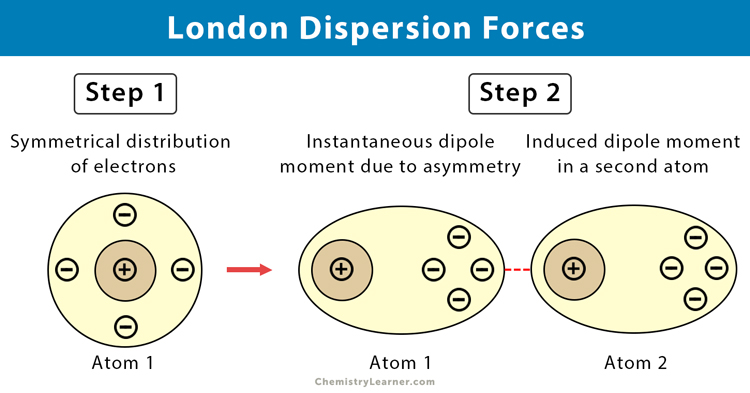
The occurrence of these short-range interactions is due to the fact that any atom will, at any given instant, be likely to possess a finite dipole moment as a result of the movement of electrons around the nuclei. In una molecola non polare gli elettroni si muovono costantemente, a un certo punto in modo casuale, per lo più si trovano verso unestremità di una molecola creando un dipolo istantaneo. We recast the description of London’s dispersion interaction in modern terminology and show that dispersion is caused by the correlation of electrons on different molecules and is hence attractive at all intermolecular separations. Van der Waals or London dispersion forces are the universal forces responsible for attractive interactions between nonpolar molecules. Dipoli indotti da dipolo istantaneo La bacheca che insegno è appena passata dalluso del termine dipoli dipolo-indotto istantaneo alluso delle forze di Londra, e il passaggio non ha senso. London’s description of dispersion is strictly only valid at large intermolecular separations since it is predicated on the free mutual rotation of molecules. Lontoon dispersiovoimat johtuvat ohimenevistä dipoleista, jotka esiintyvät elektronien pilvien välillä. This communication presents a first-principles method for computing the dispersion interaction quantum chemically. London dispersion forces happen in all molecules whether nonpolar or polar, and nonpolar molecules only have this force going on. Hence, it is of interest to be able to quantify dispersion when considering intermolecular potentials using quantum chemical techniques. Dispersion affects crystallisation, self-assembly, enzyme selectivity, and surface interactions, among countless other processes. Dispersion is a ubiquitous intermolecular force that affects short- and long-range potentials between molecules.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
